About our Company

Excellence In Complex Molecule Manufacturing Cutting-Edge Facilities & Technologies
Kwality Pharmaceuticals Ltd. stands at the forefront of innovation in pharmaceutical manufacturing, with proven expertise in developing and producing complex molecules using state-of-the-art technologies. Our advanced infrastructure and unwavering commitment to research and development position us as a trusted partner for global pharmaceutical requirements.
Recognized Globally for Quality
We are proud to be among the fastest pharmaceutical manufacturing company for complex molecules in India. Our consistent delivery of high-quality formulations has earned us certifications and approvals from global regulatory authorities including WHO-GMP, EUGMP, PIC/s (Anvisa, SFDA), INVIMA (Colombia), COFEPRIS (Mexico), and DIGEMID (Peru), among others. These achievements are a testament to our unwavering commitment to excellence and compliance with international quality standards.

Our vision is to be a leading pharmaceutical company in India and to become a significant global player by providing high quality, affordable and innovative solutions in medicine and treatment.
Our mission is to create powerful new therapies that address real health problems.
- 1. We are committed to manufacture quality products that are economically viable for our customers.
- 2. Providing value to our investors, all within a supportive environment.
- 3. Rewarding the work environment for our employees.
- 4. We are responsible to society. We shall be good corporate citizens and will be driven by high ethical standards in our practices.
Mr.Ramesh Arora
(Managing Director)

Ramesh Arora is a highly regarded figure in the pharmaceutical industry with an extensive experience. His visionary leadership has propelled Kwality Pharma Ltd to new heights, establishing it as a trusted name in the international market. Today, Ramesh Arora guides the company on strategic decision-making for the company’s growth along with grooming the next generation of management. His strategic inputs and relationship managements with various stake holders have helped the company be ahead in a competitive industry. He also plays a important role in building internal technology and exploring new business opportunities.
As the director at Kwality Pharma, Mr. Ajay Arora assumes a crucial role in overseeing the company's manufacturing planning and operations. With a B. Pharm. degree and more than 20 years of experience, his contribution to organizational development is of paramount importance. Primarily, he takes charge of procurement of raw material including API, ensuring the acquisition of machinery and other essential requirements. Additionally, he actively manages day-to-day manufacturing activities and provides oversight to various departments, including conducting initial audits of documentation, production, and inspections. Ajay Arora's extensive knowledge of pharmaceutical processes and plant-level engineering design proves invaluable when establishing multiple plants efficiently within the company. His entrepreneurial spirit and technocratic mindset further enhance his capabilities.
Mr.Ajay Arora
(Director)

Mr.Aditya Arora
(Director)

Aditya Arora is a dynamic and highly motivated leader who has quickly grasped the intricacies of the pharmaceutical business. Despite being a commerce graduate, his level of attention to detail and understanding surpasses expectations, often leading industry professionals to mistake him for a formal pharmaceutical expert. During his early years at the company, Aditya took on various roles encompassing QA, QC, and manufacturing processes. Recognizing the potential in both semi-regulated and regulated markets, he has now taken charge of spearheading the organization's transition towards regulated markets. His enthusiasm and comprehensive understanding of regulatory requirements across different countries and regions have been instrumental in establishing new plants and seizing new opportunities for the company. Currently, Aditya is actively involved in all aspects of the company, including production, quality assurance, quality control, and regulatory filings. His efforts are focused on shaping the organization for the next phase of growth, as he navigates Kwality towards a prosperous future
Mrs. Anu Arora joined the company 12 years back and makes a powerful impact and justify the need for women to be at the forefront. She heads the company's HR department, deals with various humanitarian issues, and collaborates with organizational administration. Completely Adaptable and dependable, with her Advanced interpersonal skills and a knack for developing rapport and building relationships, she can drive changes towards the betterment of all people associated with the company.
Mrs.Anu Arora
(Director)

Cutting-Edge Facilities & Technologies
Our manufacturing plants are equipped with top tier equipment, automation systems, and advanced techniques that align with regulations from various high regulated authorities along with SCHEDULE M and WHO norms. Every aspect of production is guided by modern practices and stringent quality checks, ensuring efficiency, accuracy, and safety at every stage of the process.
Certified for Quality, Safety & Environmental Compliance
Kwality Pharmaceuticals is ISO 9001:2015 and ISO 14001:2015 certified, reflecting our dedication to quality management systems and environmental sustainability. We don’t just meet regulatory benchmarks, we aim to exceed them. Our facilities are built to reduce waste, optimize resources, and ensure safe working conditions, forming the backbone of our responsible manufacturing approach.
Robust Quality Assurance Systems
Every raw material input used in our manufacturing process is meticulously recorded and tested as per standard operating procedures (SOPs). In-process testing is carried out at every stage of production to maintain product consistency and compliance. These stringent controls ensure that each product meets the highest safety and efficacy standards before reaching patients across the globe.
Strong Global Presence
We have a growing international footprint, supplying high-quality finished formulations to various countries across Europe, Latin America, Africa, Mena, GCC, Russia, Asia, and the CIS region. Our presence in regulated and semi-regulated markets showcases our adaptability to diverse compliance environments and market demands.
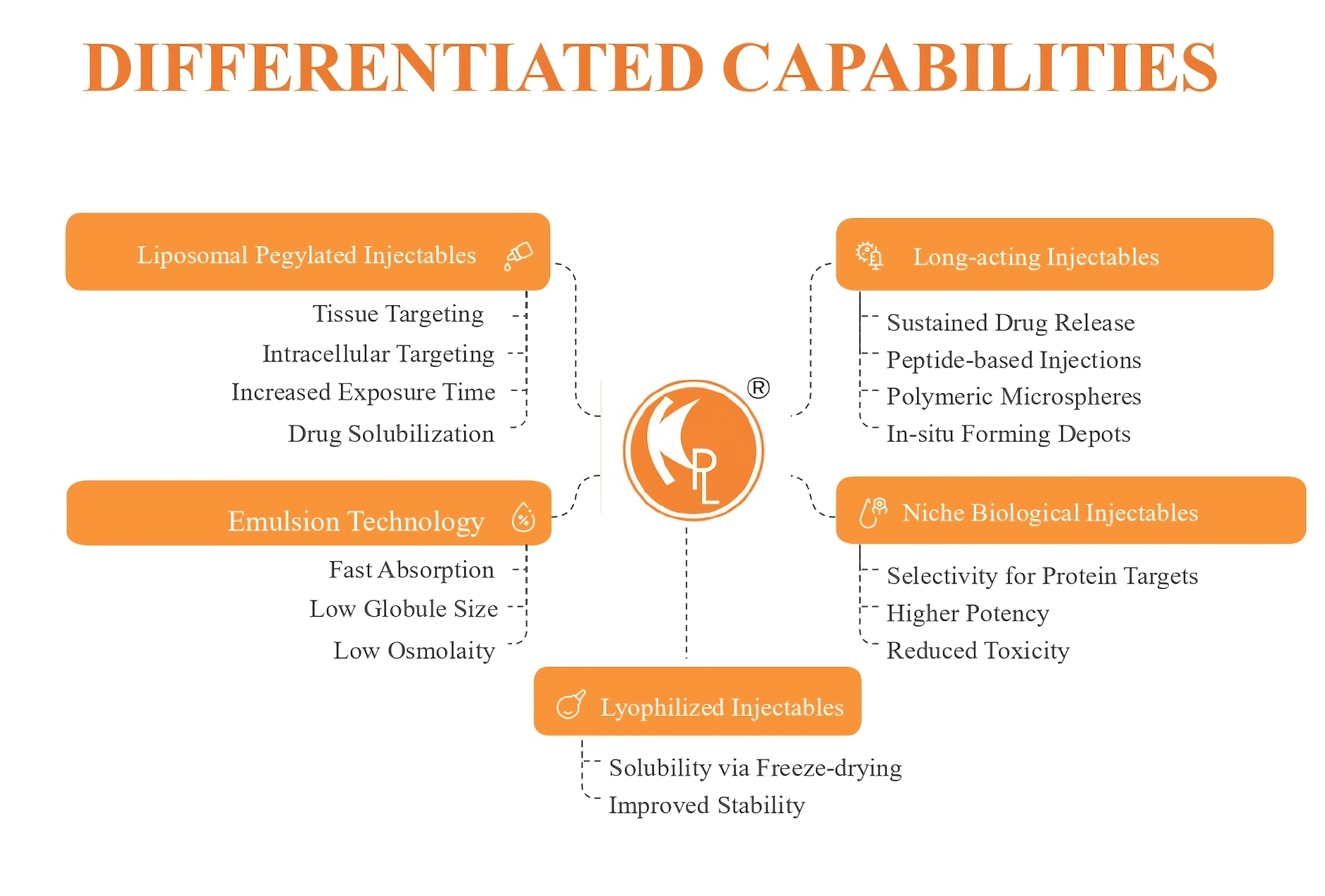
Driving Innovation for the Future
Innovation is the core of our mission. We are continuously developing new products and expanding our therapeutic portfolio to meet emerging healthcare needs. Our research team is engaged in the development of complex generics and niche products that aim to provide affordable yet advanced treatment solutions worldwide.
A Trusted Name in Pharma
With a legacy that dates back to 1983, Kwality Pharmaceuticals has evolved into a name synonymous with trust, quality, and global competence. Headquartered in Amritsar, India, and supported by our second campus in Himachal Pradesh, we remain committed to delivering better health outcomes through pharmaceutical excellence.

Amritsar Manufacturing Facility
Strategically located in Punjab, our Amritsar plant is equipped with state-of-the-art infrastructure to meet global quality standards.

Corporate Head Office
Our central command hub for strategic decisions, administration, and global operations.

Himachal Pradesh Unit
Surrounded by serene hills, this eco-friendly unit ensures clean production aligned with global quality standards.